PROJECT OVERVIEW
PROJECT DETAILS
- Research Name Cracking
- Category Process
- Location Vilnius, Lithuania

Ultrasonic Molecular Lightening: The Physics of "Cold" Cracking in the Laboratory
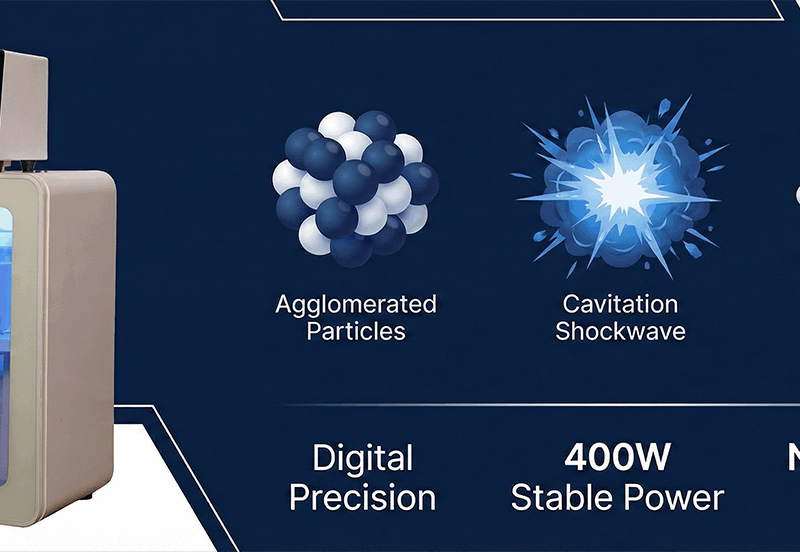
- Mechanism: Ultrasonic cracking utilizes the extreme energy of acoustic cavitation (Hot Spots: ~5,000 K, >1,000 atm) to fracture chemical bonds and reduce molecular weight without bulk thermal degradation1.
- Application: Ideal for viscosity reduction in heavy oils, polymer chain scission, and the degradation of volatile organic pollutants in wastewater research.
- The Topsonics Edge: Unlike traditional thermal cracking which is chaotic, our digital 400W system provides precise frequency locking, ensuring the exact energy density required for reproducible bond breaking.

1. Introduction: Beyond Mechanical Shearing
In the realm of material processing, a common misconception is that ultrasound is merely a "stronger mixer." While true for simple dispersions, this definition fails to capture the quantum leap in energy interaction that occurs during Molecular Lightening (or Cracking). Unlike rotor-stator mixers that rely on macroscopic turbulence and shear forces2, high-intensity ultrasound introduces a distinct state of matter interaction known as sonochemistry3.
For researchers in petrochemistry, polymer science, and environmental engineering, the challenge is often reducing the molecular weight of long-chain hydrocarbons or polymers without subjecting the bulk material to damaging high temperatures. This process, traditionally requiring high-energy thermal cracking towers, can now be simulated and studied on the benchtop using controlled acoustic cavitation.
1.1 The Acoustic Imperative
The utilization of ultrasound represents a shift from mechanical energy to microscopic energy manipulation4. When high-frequency sound waves (20 kHz) propagate through a liquid, they create alternating cycles of compression and rarefaction. If the acoustic pressure amplitude (PA) is sufficient to overcome the tensile strength of the liquid, vacuum bubbles form5.
It is the life cycle of these bubbles—specifically their violent collapse—that drives molecular lightening. This is not simple agitation; it is the generation of a micro-reactor capable of breaking carbon-carbon bonds.
Note on Terminology: While we discuss "cracking," this article focuses on sonolytic cracking (using sound energy) rather than thermal or catalytic cracking, although ultrasound can significantly enhance catalytic processes (see our article on Sonochemistry).
2. Theoretical Foundations: The Physics of Bond Breaking
To understand how a 400W laboratory device can fracture molecular chains, we must look at the Hot Spot Theory established by Suslick and others6.
2.1 The Micro-Reactor Environment
The collapse of a cavitation bubble is a near-adiabatic event. The gas trapped inside the bubble is compressed so rapidly that heat cannot dissipate into the surrounding liquid7. This results in conditions that rival the surface of the sun, yet exist only for microseconds on a microscopic scale:
- Temperature: ~5,000 K8.
- Pressure: >1,000 atm9.
- Cooling Rate: > 1010 K/s10.

2.2 Mechanism of Action by Zone
The molecular lightening occurs through two distinct mechanisms depending on the nature of the target molecule11:
Zone 1 (The Cavity Interior):
For volatile hydrocarbons or organic solvents that vaporize into the bubble, the mechanism is thermal pyrolysis. The extreme temperature inside the bubble causes direct homolytic fission of chemical bonds. Water vapor, for instance, splits into highly reactive H• and •OH radicals12. Heavy volatile organics are essentially "cracked" into lighter fractions and radicals.
Zone 2 (The Interfacial Shell):
For non-volatile long-chain polymers (like polyacrylamide or heavy crude oil fractions) that cannot enter the bubble, the mechanism is physical/mechanical. The collapse of the bubble generates shock waves and high-velocity micro-jets. These forces stretch the polymer chains coiled in the bulk liquid. When the hydrodynamic force exceeds the bond energy of the polymer backbone, the chain snaps (scission)13.
2.3 The Role of Frequency
The choice of frequency is critical. The Keller-Miksis formulation reveals that lower frequencies (like 20 kHz, used in Topsonics systems) generate larger bubbles (Rmax)14. These larger bubbles collapse with higher potential energy, favoring the physical shear forces required to break long polymer chains15. Higher frequencies (>400 kHz) are less effective for physical scission but better for radical generation16.
3. The Gap: Why Traditional Methods Fail in the Lab
Researchers attempting to study molecular degradation often face a "reproducibility crisis" when using conventional equipment.
3.1 The Thermal Trap
Traditional thermal cracking requires heating the entire bulk liquid to high temperatures. This is energy-inefficient and, more importantly, "messy" for research. It causes non-selective degradation, oxidation, and discoloration (coking) of the sample.
The Ultrasonic Alternative: The bulk liquid remains at ambient temperature (Zone 3)17. The "heat" is confined to the cavitation bubbles. This allows for "cold cracking"—reducing viscosity or molecular weight without burning the sample.
3.2 The Analog/Mechanical Problem
Many labs still use older ultrasonic homogenizers with analog tuning knobs. As the viscosity of the fluid changes (which happens as molecules are cracked and the liquid becomes thinner), the acoustic impedance (Z) of the medium changes (Z = ρ · c)18.
The Failure: An analog system cannot adapt quickly enough. The horn falls out of resonance, and the energy delivery drops, leading to inconsistent cracking rates.
The Power Fallacy: Simply buying a high-wattage industrial probe for a lab beaker often leads to low intensity due to surface area distribution19. You need focused intensity, not just raw power.
4. The Topsonics Solution: Precision for Reaction Kinetics
At Topsonics, we engineered our 400W system to address the specific needs of kinetic studies and molecular engineering. We moved away from the "brute force" approach to a "smart resonance" approach.
4.1 Digital Frequency Tracking
As molecular lightening occurs, the fluid properties change dynamically. Our system monitors the resonance frequency of the stack (typically 20 kHz ± 100 Hz) in real-time20.
Result: The system maintains peak displacement amplitude at the probe tip regardless of viscosity changes, ensuring that the shear forces required for chain scission are constant throughout the experiment.
4.2 Integrated Temperature Control Strategy
While the cracking happens in the "hot spot," preserving the bulk sample is crucial, especially for biological polymers or volatile extracts. We recommend and support the use of jacketed flow cells21. This allows the user to act as a heat exchanger, removing the waste heat generated by shock waves while allowing the cavitation events to perform the chemical work.
4.3 Scale-Up Logic
Our lab unit is designed to predict industrial outcomes. By calculating the Specific Energy (Es), researchers can determine exactly how many Joules per milliliter are required to achieve a specific reduction in molecular weight23.
This metric allows for a seamless transition from our benchtop unit to larger flow-through reactors.
5. Applications in Industry & Research
- Polymer Depolymerization: Ultrasound is the gold standard for reducing the Polydispersity Index (PDI) of polymers. Long chains are preferentially broken in the center, resulting in a sharp narrowing of the molecular weight distribution.
- Heavy Oil Upgrading (Visbreaking): In the petrochemical sector, reducing the viscosity of heavy crude oil is vital for transport. Ultrasound cracks the long-chain asphaltenes and paraffins.
- Wastewater Remediation: Molecular cracking extends to the destruction of pollutants. The Weissler Effect (production of •OH radicals)25 attacks organic dyes and pharmaceuticals.
6. Comparative Data: Thermal vs. Ultrasonic
The following table and visualization highlight the operational differences between traditional thermal cracking and ultrasonic molecular lightening.
| Feature | Thermal Cracking | Ultrasonic Cracking (Topsonics) |
|---|---|---|
| Energy Source | External Bulk Heating | Internal Cavitation Bubbles |
| Bulk Temperature | High (>300°C) | Ambient / Controlled |
| Reaction Time |
Hours
High |
Minutes26
Low |
| Selectivity | Low (Random scission) | High (Center-chain scission) |
| Sample Integrity | Risk of charring/coking | Preserved |

7. FAQ: Expert Insights
Q: Can a 400W lab unit really "crack" oil?
A: Yes, on a research scale. While you won't refine barrels of oil, a 400W unit generates the same energy density (W/cm²) at the probe tip as an industrial unit. This allows you to define the kinetics and feasibility of the process before scaling up27.
Q: Does this work for all polymers?
A: It is most effective for high molecular weight polymers dissolved in a solvent. The effect diminishes as the molecular weight drops below a certain threshold (limiting molecular weight), as the chains become too short to be caught in the shear field28.
8. Conclusion
Molecular lightening via ultrasound is a precise, non-thermal method for modifying the structure of matter. Whether you are reducing the viscosity of a bio-fuel or studying the degradation of environmental pollutants, the key is controlling the cavitation event. At Topsonics, we don't just sell a generator; we provide the acoustic precision required to turn a chaotic bubble collapse into a reliable scientific tool.
9. References
- The utilization of high-intensity ultrasound in fluid processing represents a paradigm shift...
- Unlike conventional homogenization techniques... ultrasonic homogenization harnesses the stochastic and violent power of acoustic cavitation.
- This phenomenon... generates energy densities on a microscopic scale that are otherwise impossible to achieve...
- ...ultrasonic processing is not merely a method of mixing, but a distinct state of matter interaction—often termed "sonochemistry"...
- When these waves propagate through a liquid medium, they induce vibrational motion...
- The classical description of a single spherical bubble... is provided by the Rayleigh-Plesset equation.
- The consensus mechanism for sonochemistry... is the Hot Spot Theory.
- Spectroscopic analysis of sonoluminescence... reveals effective temperatures of ~5,000 K.
- This implies heating and cooling rates in excess of 1010 K/s.
- Suslick identifies three distinct zones...
- Water vapor splits into H• and •OH radicals...
- It is affected primarily by the shock waves (shear forces)...
- Designing a horn requires ensuring that its longitudinal resonance frequency matches...
- Scale-up is calculated based on Specific Energy...